Eligible commercially-insured patients may pay as little as

GETTING STARTED WITH
LUMIGAN® 0.01%
Remember to use LUMIGAN® 0.01% once a day, every day.
The recommended dose of LUMIGAN® 0.01% is 1 drop in the affected eye(s) once daily in the evening.
LUMIGAN® 0.01% may be used alone or with other eye drops to lower eye pressure for patients with open-angle glaucoma or ocular hypertension.
If you use more than 1 type of topical eye drop, they should be applied at least 5 minutes apart.
Instructions for applying eye drops
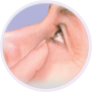
Step 1
Wash your hands. Tilt your head back and look at the ceiling.
- If you wear contact lenses, remove them first, then wait 15 minutes after using your eye drops to put them back into your eyes.
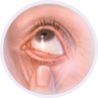
Step 2
Using your index finger, pull down your lower eyelid to form a pocket.
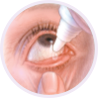
Step 3
Gently squeeze 1 drop into the pocket. Don’t let the bottle tip touch your eye, your fingers, or anything else.
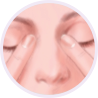
Step 4
Gently close your eyes and lightly press on the inside corners of your eyes.
Step 5
Carefully blot away any excess liquid that may be on your skin.
USE
LUMIGAN® (bimatoprost ophthalmic solution) 0.01% is used for the reduction of high eye pressure, also called intraocular pressure (IOP), in people with open-angle glaucoma or ocular hypertension.
IMPORTANT SAFETY INFORMATION
Do not use LUMIGAN® 0.01% if you are allergic to any of the ingredients.
LUMIGAN® 0.01% can cause increased brown coloring of the iris, which may be permanent. LUMIGAN® 0.01% can cause darkening of the eyelid skin and eyelashes, which may be reversible after treatment is stopped. The long-term effects of the increased dark coloring are not known.
LUMIGAN® 0.01% may slowly increase the growth and thickness of eyelashes, which is usually reversible after treatment is stopped.
Prostaglandin analogs, including bimatoprost (the active ingredient in LUMIGAN® 0.01%), have been reported to cause inflammation inside the eye and may make existing inflammation inside the eye worse.
Macular edema (swelling of the macula, the central part of the retina), including cystoid macular edema, has been reported during treatment with bimatoprost ophthalmic solution. Use caution if you use LUMIGAN® 0.01% and you don’t have your natural lens, have a torn posterior lens capsule and an artificial lens implant, or have a known risk factor for macular edema.
Avoid allowing the tip of the dispensing bottle to touch the eye, anything around the eye, fingers, or any other surface to avoid contamination by common bacteria known to cause eye infections (bacterial keratitis). Using contaminated solutions can cause serious damage to the eye and loss of vision.
If you have eye surgery, eye trauma, or infection, or develop any eye reactions, immediately consult with your physician about continuing the use of LUMIGAN® 0.01%.
LUMIGAN® 0.01% contains an ingredient that may be absorbed by and cause discoloration of soft contact lenses. If you wear contact lenses, remove them before using LUMIGAN® 0.01%. Then wait 15 minutes after using LUMIGAN® 0.01% before you put your contacts back into your eyes.
If you use more than one drug in the eye, be sure to wait at least 5 minutes between each drug application.
The most common side effect in the clinical trial was eye redness. Other side effects include eye irritation, eye pain, itchy eyes, discoloration of skin/eyelids, and blurred vision. These are not all of the possible side effects with LUMIGAN® 0.01%. Tell your doctor if you have any side effect that bothers you or that does not go away.
Please see full Prescribing Information for LUMIGAN® at
https://www.rxabbvie.com/pdf/
lumigan_pi.pdf.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.
If you are having difficulty paying for your medicine, AbbVie may be able to help. Visit AbbVie.com/myAbbVieAssist to learn more.




