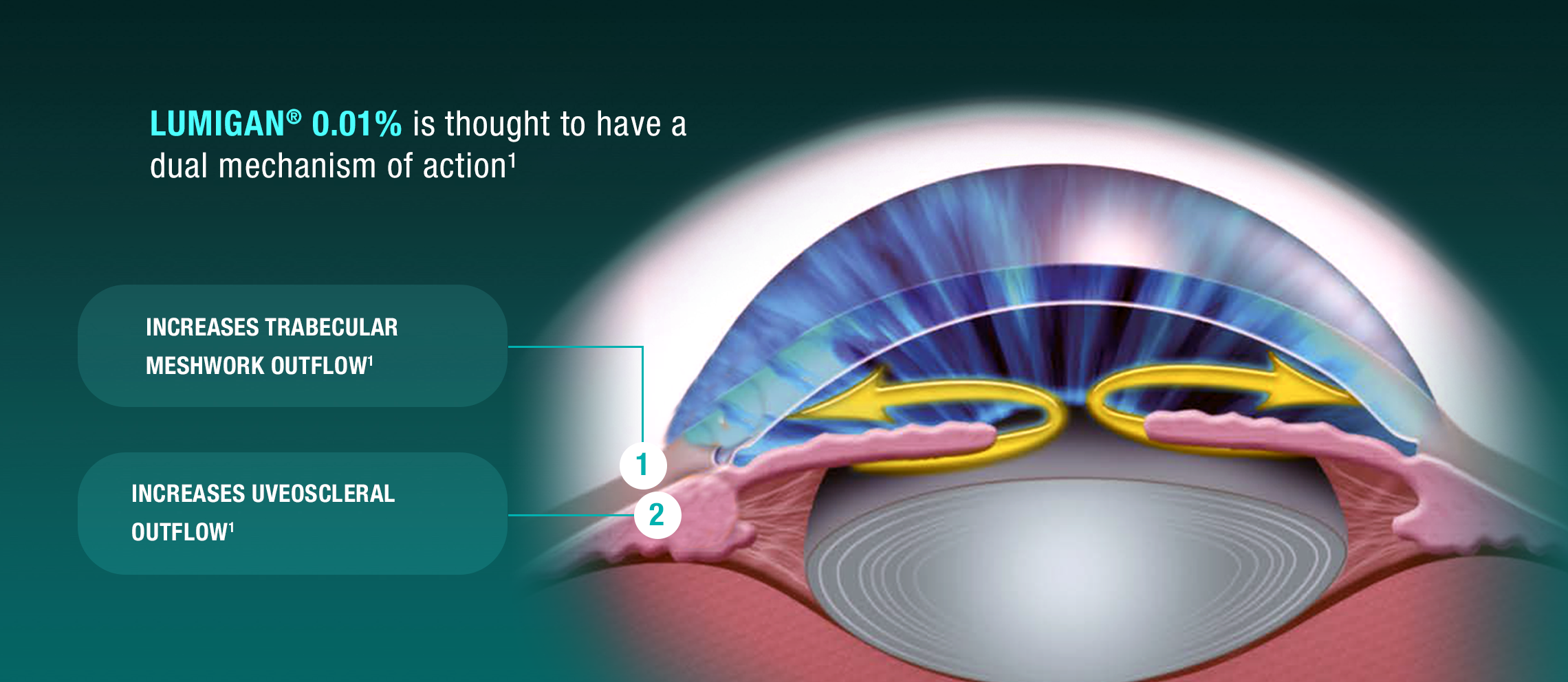
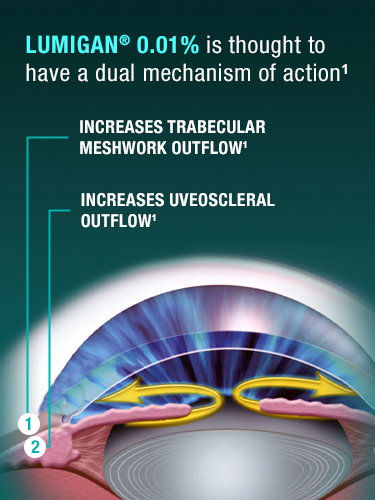
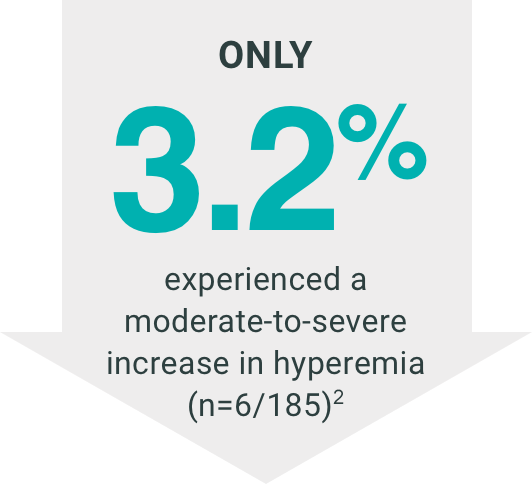
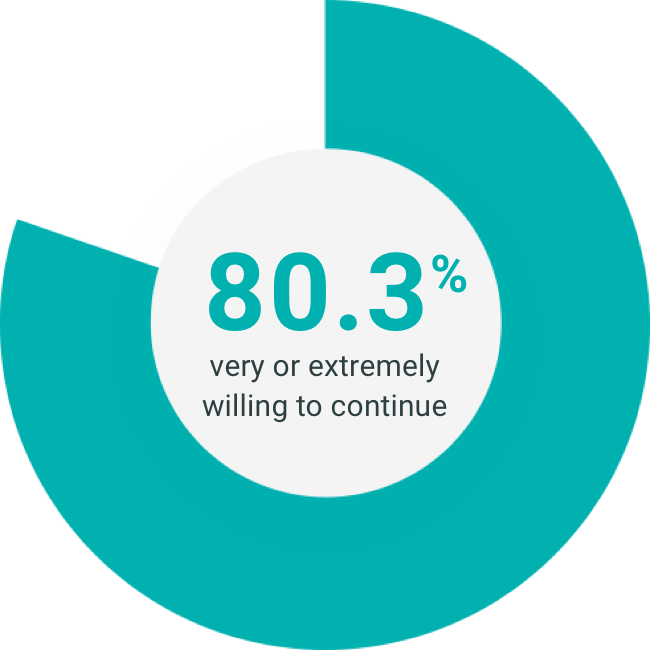
INDICATION
LUMIGAN® 0.01% (bimatoprost ophthalmic solution) is indicated for the
reduction of elevated intraocular
pressure in patients with open angle glaucoma or ocular hypertension.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
LUMIGAN® 0.01% is contraindicated in patients
with hypersensitivity to bimatoprost or to any of the ingredients.
WARNINGS AND PRECAUTIONS
Pigmentation
Bimatoprost ophthalmic solution has been reported to cause changes to pigmented tissues.
The most
frequently reported changes have been increased pigmentation of the iris, periorbital
tissue (eyelid) and
eyelashes. Pigmentation is expected to increase as long as bimatoprost is administered.
After
discontinuation of bimatoprost, pigmentation of the iris is likely to be permanent,
while pigmentation of the
periorbital tissue and eyelash changes have been reported to be reversible in some
patients. Patients
who receive treatment should be informed of the possibility of increased pigmentation.
The long term
effects of increased pigmentation are not known. Iris color change may not be noticeable
for several
months to years. While treatment with LUMIGAN®
0.01% can be continued in
patients who develop noticeably increased iris pigmentation, these patients should be
examined regularly.
Eyelash Changes
LUMIGAN® 0.01% may gradually change eyelashes
and vellus hair in the treated eye. These changes
include increased length, thickness, and number of lashes. Eyelash changes are usually
reversible upon
discontinuation of treatment.
Intraocular Inflammation
Prostaglandin analogs, including bimatoprost, have been reported to cause intraocular
inflammation. In
addition, because these products may exacerbate inflammation, caution should be used in
patients with
active intraocular inflammation (e.g., uveitis).
Macular Edema
Macular edema, including cystoid macular edema, has been reported during treatment with
bimatoprost
ophthalmic solution. LUMIGAN® 0.01% should be
used with caution in aphakic patients, in pseudophakic
patients with a torn posterior lens capsule, or in patients with known risk factors for
macular edema.
Bacterial Keratitis
There have been reports of bacterial keratitis associated with the use of multiple-dose
containers of topical
ophthalmic products.
Contact Lens Use
LUMIGAN® 0.01% contains benzalkonium chloride,
which may be absorbed by and cause discoloration of
soft contact lenses. Contact lenses should be removed prior to instillation of LUMIGAN® 0.01% and may
be reinserted 15 minutes following its administration.
ADVERSE REACTIONS
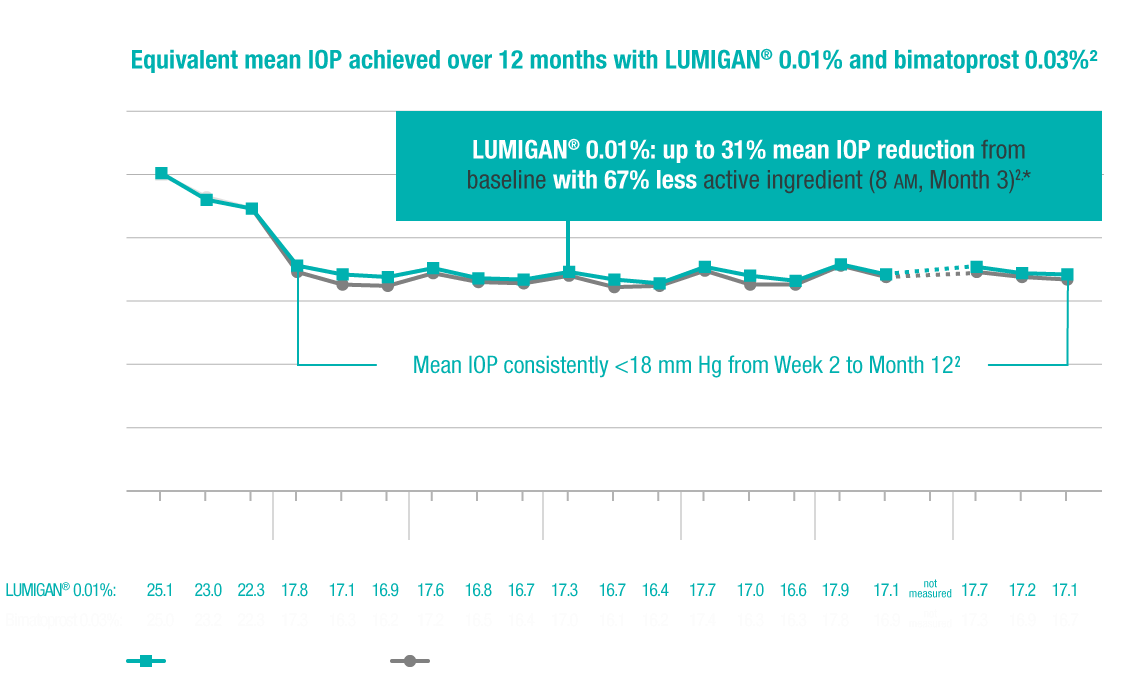
In a 12-month clinical study with bimatoprost ophthalmic solutions 0.01%, the most
common adverse
reaction was conjunctival hyperemia (31%). Approximately 1.6% of patients discontinued
therapy due to
conjunctival hyperemia. Other adverse drug reactions (reported in 1 to 4% of patients)
with LUMIGAN® 0.01%
in this study included conjunctival edema, conjunctival hemorrhage, eye irritation, eye
pain, eye
pruritus, erythema of eyelid, eyelids pruritus, growth of eyelashes, hypertrichosis,
instillation site irritation,
punctate keratitis, skin hyperpigmentation, vision blurred, and visual acuity reduced.
USE IN SPECIFIC POPULATIONS
Pediatric Use
Use in pediatric patients below the age of 16 years is not recommended because of
potential safety
concerns related to increased pigmentation following long-term chronic use.
Please see accompanying full Prescribing Information or visit https://www.rxabbvie.com/pdf/
lumigan_pi.pdf